CUPERTINO — Apple announced plans to buy back up to $110 billion in shares of its stock and also boost its dividend. The company last week reported financial results for its fiscal 2024 second quarter ended March 30, 2024. In the second quarter, revenue was $90.8 billion, down 4 percent year over year, and quarterly […]
Reddit Shares Pop on First Earnings Report
SAN FRANCISCO — Shares in online discussion forum Reddit surged over 16% Tuesday in after-hours trading when the company released first quarter earnings for 2024. This was the first earnings report for Reddit as a publicly-traded company. “It was a strong start to the year and a milestone quarter for Reddit and our communities as […]
Amazon Says Prime Delivery Speeds are Faster Than Ever
Amazon Stores CEO Doug Herrington reports the company has set new records for Prime delivery speeds in the first three months of 2024, with more than two billion items arriving the same or next day to Prime members around the world. In March, nearly 60% of Prime member orders arrived the same or next day […]
Taylor Swift Album Sets Record on Amazon Music
Taylor Swift has a “big reputation” for shattering streaming records, and her 11th studio album is no exception. THE TORTURED POETS DEPARTMENT became the most-streamed album in its first day ever on Amazon Music. THE TORTURED POETS DEPARTMENT marks Taylor Swift’s eleventh studio album, following her Midnights release in 2022 and her re-releases of Speak Now (Taylor’s Version) and 1989 (Taylor’s Version). Fans […]
Apple Introduces M4 Chip for iPad Pro
CUPERTINO — Apple has introduced M4, its most powerful chip to date and will be available in the all-new iPad Pro. Built using second-generation 3-nanometer technology, M4 is a system on a chip (SoC) that advances the industry-leading power efficiency of Apple silicon and enables the incredibly thin design of iPad Pro. It also features […]
YouTuber Emma Chamberlain to Interview Stars on Met Gala Red Carpet
Emma Chamberlain has the enviable job of interviewing celebrities arriving at the Met Gala tonight in NYC. The popular YouTuber will be doing the interviews live over streaming on Vogue.com and also airing on YouTube and TikTok channels. Chamberlain has become one of the most popular stars on YouTube with over 12 million subscribers since […]
Starbucks Has 38,951 Stores Worldwide
Starbucks Corporation, the world’s largest coffee chain, reported financial results for its 13-week fiscal second quarter ended March 31, 2024. Sales declined 4% in the quarter and the chain now has 38,951 stores around the world. Q2 Fiscal 2024 Highlights Global comparable store sales declined 4%, driven by a 6% decline in comparable transactions, partially […]
HGTV Star of Flip It to Win It Sentenced to Four Years in Jail
A former HGTV star was sentenced to four years in jail and ordered to pay back close to $10 million for committing real estate and financial fraud against 11 victims. Charles “Todd” Hill, 58, of Los Gatos, star of “Flip It to Win It”, was remanded to jail last week. The show’s concept was that […]
Walmart Launches New Private Food Brand Bettergoods:
Walmart, the nation’s largest retailer, has introduced bettergoods: a new private band that delivers quality, unique, chef-inspired food at an incredible value. Bettergoods marks Walmart’s largest private brand food launch in 20 years and the fastest food private brand Walmart has brought to market, highlighting the speed with which Walmart can bring trend and innovation […]
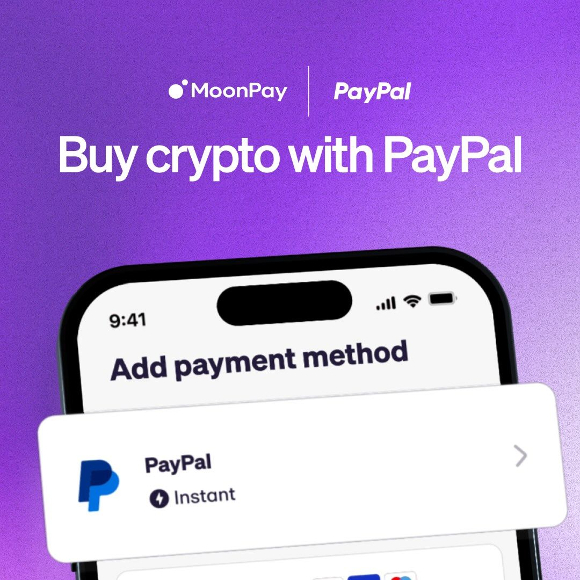
MoonPay Lets Users Buy & Sell Crypto With PayPal
MoonPay, a Web3 infrastructure company, is teaming up with PayPal to allow MoonPay users in the U.S. to seamlessly buy cryptocurrency using their PayPal account. The integration streamlines transactions for new MoonPay users who already have a PayPal account and use it for online transactions. When using PayPal, users can fund their cryptocurrency purchases by […]
Small Business Owners Worried About Inflation, Economy: CNBC/SurveyMonkey
ENGLEWOOD CLIFFS, NJ and SAN MATEO, CA — Business network CNBC and SurveyMonkey announced the results of their quarterly CNBC|SurveyMonkey Small Business Survey. After a disappointing March inflation report, the survey, which polled over 2,000 small business owners in the U.S. from April 8-12, 2024, finds that optimism over inflation has dropped among small business […]
San Francisco Woman Convicted of Stealing $60,000 From Target
SAN FRANCISCO – San Francisco District Attorney Brooke Jenkins announced her office has secured a conviction of Aziza Graves (43), of San Francisco, after a trial by jury for repeated theft from retailers. Ms. Graves was convicted of one felony count of grand theft in violation (PC 487(a)) and 52 misdemeanor counts of petty theft […]